A New Mechanistic Approach to
Migraine Management
A patent applied synergistic formulation targeting novel molecular pathways in migraine pathophysiology.
Innovative Molecular Targeting
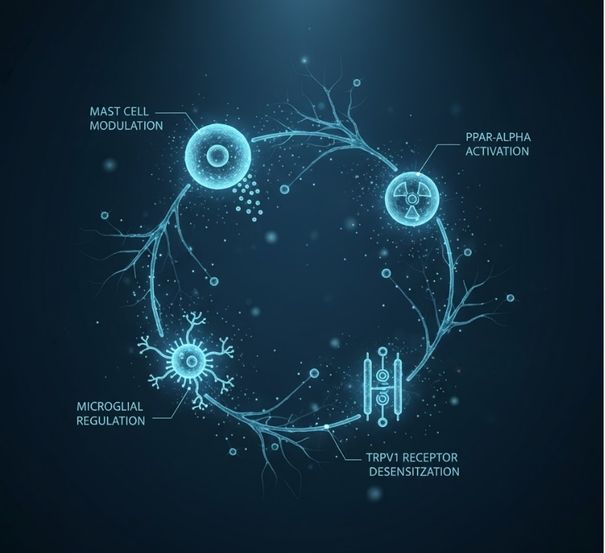
CephaMig™ modulates previously unaddressed molecular pathways including mast cells, microglia, PPAR-α, and TRPV1 receptors—bridging the gap left by conventional migraine therapies.
Clinically Proven Efficacy
Demonstrated reduction in migraine frequency, intensity, and duration with decreased dependence on rescue medication.
Safe and Well-Tolerated
Composed of clinically validated ingredients with proven safety across multiple studies and regulatory frameworks.
Science Meets Precision
CephaMig™ represents the evolution of migraine management—where neuroscience, molecular pharmacology, and clinical evidence converge.
Our research-driven approach identifies and modulates critical pathways in neuroinflammation, improving patient outcomes without adverse effects.
Key Focus Areas
- Neuroinflammation and mast cell modulation
- PPAR-α and TRPV1 receptor targeting
- Enhanced neural stability and reduced pain signaling
- Complementary to conventional antimigraine therapies
“CephaMig™ Defining a New Era in Migraine Science”
Developed through years of clinical research, CephaMig™ stands as a patent applied international innovation for migraine prophylaxis.
Discover the Science Behind CephaMig™
CephaMig™ is the result of advanced research integrating palmitoylethanolamide (PEA), Ginkgolide-B, and Magnesium—a synergistic composition designed to optimize neuronal balance and reduce migraine burden.
Palmitoylethanolamide (PEA)
Targets neuroinflammation and stabilizes mast cell activity to prevent migraine initiation.
Ginkgolide-B
Inhibits platelet-activating factor (PAF), reducing vascular instability and aura symptoms.
Magnesium
Supports neuronal energy balance and mitigates excitotoxicity associated with migraine attacks.
Recognized by Global Experts
“CephaMig™ represents a significant advancement in migraine prevention through multi-target molecular modulation.”
— Clinical Investigator
From Research to Real-World Impact
By modulating neuroinflammatory and neurosensory pathways, CephaMig™ achieves multifactorial migraine prevention. It offers neuroprotection, reduced attack frequency, and enhanced quality of life through a validated, multimodal novel molecular target based mechanism.